Breyanzi® is THE ONE studied in a broad range of patients, including older patients with moderate comorbidities, as well as young and fit patients1-5

In R/R LBCL trials, response rates were observed across patient subgroups, regardless of age, fitness status, response to prior therapy, or receipt of bridging therapy2,4,6,7
*Patient ages in TRANSFORM and PILOT ranged from 20 to 75 years and 53 to 84 years, respectively.1
2L, second-line; LBCL, large B-cell lymphoma; R/R, relapsed or refractory.
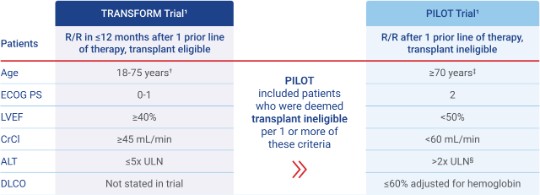
In the 3L+ TRANSCEND trial, patients were refractory or relapsed after ≥2 prior therapies, ≥18 years old, ECOG PS ≤2, LVEF ≥40%, CrCl ≥30 mL/min, and ALT ≤5x ULN.1
Identify eligible patients early and connect them with a CAR T-cell therapy treatment center
†Median age was 59 years (range: 20-75 years).1
‡Eligible patients for the PILOT trial were required to be ≥18 years old. Median age was 74 years (range: 53-84 years).1,3
§PILOT criteria included ALT/AST.1
3L, third-line; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CAR, chimeric antigen receptor; CrCl, creatinine clearance; DLCO, diffusing capacity of the lungs for carbon monoxide; ECOG PS, Eastern Cooperative Oncology Group performance status; LVEF, left ventricular ejection fraction; ULN, upper limit of normal.
Choose Breyanzi, THE ONE CAR T for the broadest range of patients with 2L LBCL1
References
- Breyanzi [package insert]. Summit, NJ: Bristol-Myers Squibb Company; 2025.
- Kamdar M, Solomon S, Arnason J, et al. Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim analysis of an open-label, randomised, phase 3 trial. Lancet. 2022;399(10343):2294-2308.
- Sehgal A, Hoda D, Riedell PA, et al. Lisocabtagene maraleucel as second-line therapy in adults with relapsed or refractory large B-cell lymphoma who were not intended for haematopoietic stem cell transplantation (PILOT): an open-label, phase 2 study. Lancet Oncol. 2022;23(8):1066-1077. doi:10.1016/S1470-2045(22)00339-4
- Crombie JL, Nastoupil LJ, Andreadis C, et al. Multicenter, real-world study in patients with relapsed or refractory large B-cell lymphoma who received lisocabtagene maraleucel in the United States. Presented at: American Society of Hematology Annual Meeting & Exposition; December 9, 2023; San Diego, CA. Presentation 104.
- CADTH Reimbursement Review. Lisocabtagene maraleucel (Breyanzi). Can J Health Tech. 2022;2(10).
- Nastoupil L, Kamdar M, Chavez JC, et al. Subgroup analyses of primary refractory (refr) vs early relapsed (rel) large B-cell lymhpoma (LBCL) from the study of lisocabtagene maraleucel (liso-cel) vs standard of care (SOC) as second-line (2L) therapy. Presented at: American Society of Clinical Oncology Annual Meeting; June 2-6, 2023; Chicago, IL.
- Abramson J, Solomon S, Arnason J, et al. Lisocabtagene maraleucel as second-line therapy for large B-cell lymphoma: primary analysis of the phase 3 TRANSFORM study. Supplementary appendix. Blood. 2023;141(14):1675-1684.
- Chen AI, Maziarz RT. Can we truly define 'fitness' for CAR-T therapy in large B cell lymphoma patients? Chin Clin Oncol. 2024;13(1):15. doi:10.21037/cco-23-78






