| Age (N=68)1 | |
|---|---|
| Median age, years (range) | 69 (36-86) |
| Prior therapies (N=68)1 | |
| Median number of prior therapies (range) | 3 (2-11) |
| Prior BTKi | 100% |
| Refractory§ to prior BTKi | 56% |
| Prior stem cell transplantation | 32% |
| High-risk features (N=68)1,3 | |
| Refractory to last therapy∥ | 69% |
| High-risk feature | |
| Ki67 ≥30% | 77% |
| Complex karyotype | 31% |
| Blastoid morphology | 29% |
| TP53 mutation | 25% |
| CNS involvement | 10% |
An open-label, multicenter, single-arm trial
Breyanzi®: THE ONE evaluated in adult patients with R/R MCL who have received at least 2 prior lines of systemic therapy, including a BTKi1
Primary endpoints: ORR by an IRC and safety1,2
Select secondary endpoints: CR, DOR, PFS, and OS2
Bridging chemotherapy was permitted between leukapheresis and lymphodepleting chemotherapy.1
Breyanzi provides deep and durable complete responses in a one-time infusion1*
Response rates (N=68)1†

Overall response rates by mutational or proliferative status (subgroup analysis)3
Subgroup analysis limitations:
- These analyses are exploratory in nature, and definitive conclusions should not be drawn
- Numbers may not be available in the Prescribing Information

Overall response rates by prespecified subgroups3‡

Power to deliver durable response in R/R MCL1
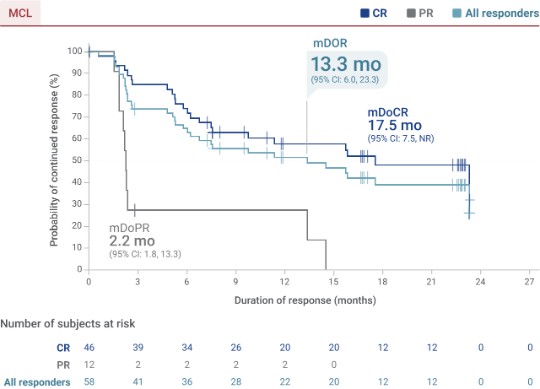
Duration of response in TRANSCEND MCL Cohort (58/68)1,3§||
Median follow-up: 22.2 months (95% CI: 16.7, 22.8)
1-month median time to first response (range: 0.7-3 months)1
*Treatment process includes leukapheresis, manufacturing, administration, and adverse event monitoring.1
†Per the 2014 Lugano classification (including bone marrow biopsy assessments), as assessed by an IRC. ORR was defined as the percentage of patients with BOR of either CR or PR after Breyanzi infusion, as determined by an IRC using 2014 Lugano classification.1
‡2-sided 95% exact Clopper-Pearson CIs.3
§Per the 2014 Lugano classification (including bone marrow biopsy assessments), as assessed by an IRC.1
∥Kaplan-Meier method was used to obtain 2-sided 95% CIs.1
BOR, best overall response; BTKi, Bruton tyrosine kinase inhibitor; CI, confidence interval; CNS, central nervous system; CR, complete response; DOR, duration of response; HSCT, hematopoietic stem cell transplantation; IRC, Independent Review Committee; MCL, mantle cell lymphoma; mDoCR, median duration of complete response; mDoPR, median duration of partial response; mDOR, median duration of response; NR, not reached; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PR, partial response; R/R, relapsed or refractory; TP53, tumor protein 53.
AN OPEN-LABEL, MULTICENTER, SINGLE-ARM TRIAL
Choose Breyanzi: THE ONE for adults with R/R MCL who have received at least 2 prior lines of systemic therapy, including a BTKi1

Screening1,2*
Broad enrollment criteria:
- Adult patients with R/R MCL who received ≥2 lines of systemic therapy, including a BTKi
- Age ≥18 years
- ECOG PS ≤1
- LVEF ≥40%
- CrCl >30 mL/min
- ALT ≤5x ULN
- PET-positive MCL with confirmed tissue diagnosis
- Secondary CNS lymphoma or prior HSCT allowed
- Adequate bone marrow†
Enrollment and leukapheresis (N=89)


Breyanzi manufacturing1
Bridging chemotherapy was permitted between leukapheresis and lymphodepleting chemotherapy; 65% (44/68) received bridging therapy


Lymphodepletion1‡
FLU 30 mg/m2 and CY 300 mg/m2 × 3 days
Patients treated with Breyanzi (68/89)


Breyanzi infusion1
2 to 7 days after FLU/CY, planned dose of 100 × 106 CAR+ T cells
- Primary endpoints: ORR (per the 2014 Lugano classification, including bone marrow biopsy assessments, as assessed by an IRC) and safety1,2
- Select secondary endpoints: CR, DOR, PFS, and OS2
Of 89 patients who underwent leukapheresis, 71 received Breyanzi and the median dose administered was 99.8 × 106 CAR-positive viable T cells (range: 90 to 103 × 106 CAR-positive viable T cells).1
The primary efficacy analysis included a total of 68 patients with MCL who received at least 2 prior lines of therapy including a BTKi, had PET-positive disease at study baseline or after bridging therapy, received conforming product in the intended dose range, and had at least 6 months of follow-up from the date of first response.1
Studied in patients you are likely to see in your practice
Broad eligibility criteria in TRANSCEND MCL Cohort make Breyanzi accessible to more patients.1
- 15% (10/68) of patients were treated in an outpatient setting1
- Bridging therapy is an option; 65% (44/68) received bridging therapy1
*Additional eligibility criteria applied.
†No prespecified threshold for blood counts.1
‡Measurable disease reconfirmed prior to lymphodepletion.2
§Defined as any response to prior BTKi that was less than partial response.1
∥Defined as best response of partial response, stable disease, or progressive disease to last systemic or HSCT treatment with curative intent.1
ALT, alanine aminotransferase; BTKi, Bruton tyrosine kinase inhibitor; CAR, chimeric antigen receptor; CI, confidence interval; CNS, central nervous system; CR, complete response; CrCl, creatinine clearance; CY, cyclophosphamide; DOR, duration of response; ECOG PS, Eastern Cooperative Oncology Group performance status; FLU, fludarabine; HSCT, hematopoietic stem cell transplantation; IRC, Independent Review Committee; LVEF, left ventricular ejection fraction; MCL, mantle cell lymphoma; ORR, overall response rate; OS, overall survival; PET, positron emission tomography; PFS, progression-free survival; R/R, relapsed or refractory; TP53, tumor protein 53; ULN, upper limit of normal.
References
- Breyanzi [package insert]. Summit, NJ: Bristol-Myers Squibb Company; 2025.
- Wang M, Siddiqi T, Gordon LI, et al. Lisocabtagene maraleucel in relapsed/refractory mantle cell lymphoma: primary analysis of the mantle cell lymphoma cohort from TRANSCEND NHL 001, a phase I multicenter seamless design study. J Clin Oncol. 2024;42(10):1146-1157. doi:10.1200/JCO.23.02214
- Data on file. BMS-REF-LIS-0051. Princeton, NJ: Bristol-Myers Squibb Company; 2024.
2009-US-2600190 03/2026

